CORONAVIRUS
UK Authorizes Pfizer Coronavirus Vaccine For Emergency Use
Dec 2, 2020, 5:31 AM

LONDON, ENGLAND - DECEMBER 02: Prime Minister Boris Johnson leaves 10 Downing Street after it was announced that the UK Government has approved the Pfizer/BioNTech Covid vaccine for use fromnext week, on December 2, 2020 in London, England. (Photo by Peter Summers/Getty Images)
(Photo by Peter Summers/Getty Images)
(AP) – British officials authorized a COVID-19 vaccine for emergency use on Wednesday, greenlighting the world’s first shot against the virus that’s backed by rigorous science and taking a major step toward eventually ending the pandemic.
The go-ahead for the vaccine developed by American drugmaker Pfizer and Germany’s BioNTech comes as the virus surges again in the United States and Europe, putting pressure on hospitals and morgues in some places and forcing new rounds of restrictions that have devastated economies.
The Medicines and Healthcare Products Regulatory Agency, which licenses drugs in the U.K., recommended the vaccine could be used after it reviewed the results of clinical trials that showed the vaccine was 95% effective overall — and that it also offered significant protection for older people, among those most at risk of dying from the disease. But the vaccine remains experimental while final testing is done.
“Help is on its way,” British Health Secretary Matt Hancock told the BBC, adding that the situation would start to improve in the spring.
“We now have a vaccine. We’re the first country in the world to have one formally clinically authorized but, between now and then, we’ve got to hold on, we’ve got to hold our resolve,” he said.
Other countries aren’t far behind: Regulators in the United States and the European Union also are vetting the Pfizer shot along with a similar vaccine made by competitor Moderna Inc. British regulators also are considering another shot made by AstraZeneca and Oxford University.
Hancock said Britain expects to begin receiving the first shipment of 800,000 doses “within days,” and people will begin receiving shots as soon as the National Health Service gets the vaccine.
Doses everywhere are scarce, and initial supplies will be rationed until more is manufactured in the first several months of next year.
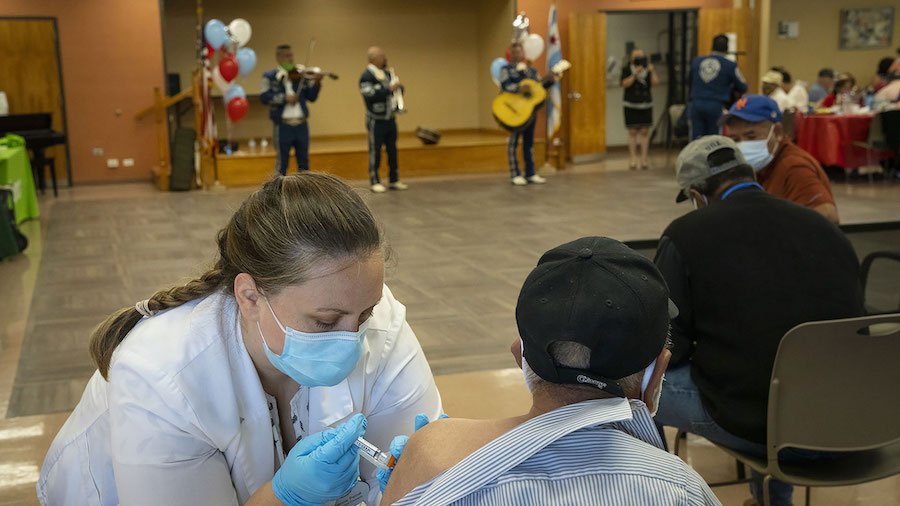
A government committee will release details of vaccination priorities later Wednesday, but Hancock said nursing home residents, people over 80, and healthcare workers and other care workers will be the first to receive the shot.
Pfizer said it would immediately begin shipping limited supplies to the U.K. — and has been gearing up for even wider distribution if given a similar nod by the U.S. Food and Drug Administration, a decision expected as early as next week.
Pfizer CEO Albert Bourla called the U.K. decision “a historic moment.”
“We are focusing on moving with the same level of urgency to safely supply a high-quality vaccine around the world,” Bourla said in a statement.
While the U.K. has ordered 40 million doses of the Pfizer vaccine, enough for 20 million people, it’s not clear how many will arrive by year’s end. Hancock said the U.K. expects to receive “millions of doses” by the end of this year, adding that the actual number will depend on how fast Pfizer can produce the vaccine.
One concern about the Pfizer-BioNTech vaccine is that it must be stored and shipped at ultra-cold temperatures of around minus 70 degrees Celsius (minus 94 degrees Fahrenheit), adding to the challenge of distributing the vaccine around the world.
Pfizer says it has developed shipping containers that use dry ice to keep the vaccine cool. GPS-enabled sensors will allow the company to track each shipment and ensure they stay cold, the company says.
“Pfizer has vast experience and expertise in cold-chain shipping and has an established infrastructure to supply the vaccine worldwide, including distribution hubs that can store vaccine doses for up to six months,” the company said in a statement.
The company also says it has agreed to work with other vaccine makers to ensure there is sufficient supply and a range of vaccines, “including those suitable for global access.”
Every country has different rules for determining when an experimental vaccine is safe and effective enough to use. Intense political pressure to be the first to roll out a rigorously scientifically tested shot colored the race in the U.S. and Britain, even as researchers pledged to cut no corners. In contrast, China and Russia have offered different vaccinations to their citizens ahead of late-stage testing.
The shots made by U.S.-based Pfizer and its German partner BioNTech were tested in tens of thousands of people. And while that study isn’t complete, early results suggest the vaccine is 95% effective at preventing mild to severe COVID-19 disease. The companies told regulators that of the first 170 infections detected in study volunteers, only eight were among people who’d received the actual vaccine and the rest had gotten a dummy shot.
“This is an extraordinarily strong protection,” Dr. Ugur Sahin, BioNTech’s CEO, recently told The Associated Press.
The companies also reported no serious side effects, although vaccine recipients may experience temporary pain and flu-like reactions immediately after injections.
Final testing must still be completed. Still to be determined is whether the Pfizer-BioNTech shots protect against people spreading the coronavirus without showing symptoms. Another question is how long protection lasts.
The vaccine also has been tested in only a small number of children, none younger than 12, and there’s no information on its effects in pregnant women.
___
Follow AP’s coverage at https://apnews.com/hub/coronavirus-pandemic and https://apnews.com/UnderstandingtheOutbreak.
___
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute’s Department of Science Education. The AP is solely responsible for all content.